Succinyl-Coa Ligase [Gdp-Forming] Alpha-Chain, Mitochondrial
(All numbering and residues are taken from first PDB file)
![]()
![]()
Bending Residue Dihedral Analysis
Residue
iResidue
i+1Distance of hinge axis to residue i in
(A) Distance of hinge axis to residue i in
(A) Change in
(deg) Change in
(deg) Angle of psi(i) axis to hinge axis
(deg) Angle of psi(i) axis to hinge axis
(deg) Percentage Progress
ALA-247
GLY-248
9.9
10.2
0.1
-6.7
70.7
68.0
-1.1
GLY-248
LEU-249
7.1
7.6
-21.2
4.3
102.3
97.0
-6.7
LEU-249
THR-250
5.7
6.1
11.3
6.3
22.7
14.6
19.7
THR-250
ALA-251
5.2
6.3
-14.4
5.2
98.7
111.5
-4.7
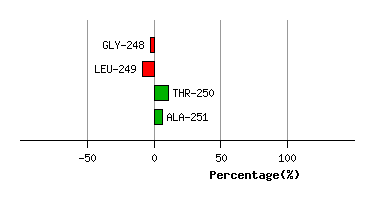
Graph shows rotational transition at bending residues and can be used
to identify hinge bending residues.
Probably only informative for interdomain rotations greater than 20 degrees
Residue
iResidue
i+1Distance of hinge axis to residue i in
(A) Distance of hinge axis to residue i in
(A) Change in
(deg) Change in
(deg) Angle of psi(i) axis to hinge axis
(deg) Angle of psi(i) axis to hinge axis
(deg) Percentage Progress
ALA-271
LYS-272
3.5
3.8
-86.0
18.3
113.1
125.9
-59.8
LYS-272
GLU-273
4.5
5.5
5.2
-2.0
19.7
21.5
2.2
GLU-273
LYS-274
3.3
3.5
-18.2
-0.4
132.7
127.8
-19.5
LYS-274
ILE-275
3.5
3.6
6.6
-2.3
80.4
83.1
-9.9
ILE-275
THR-276
7.1
7.2
-19.6
0.9
124.8
132.8
-29.7
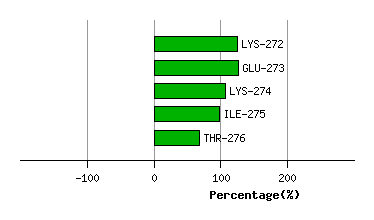
Graph shows rotational transition at bending residues and can be used
to identify hinge bending residues.
Probably only informative for interdomain rotations greater than 20 degrees